Blog Amal Fahmi – Organoide
Auswirkungen von Virusinfektionen auf die Plazenta und das Gehirn von Föten: Forschung mit Ex-vivo-Modellen und 3D-Organoiden
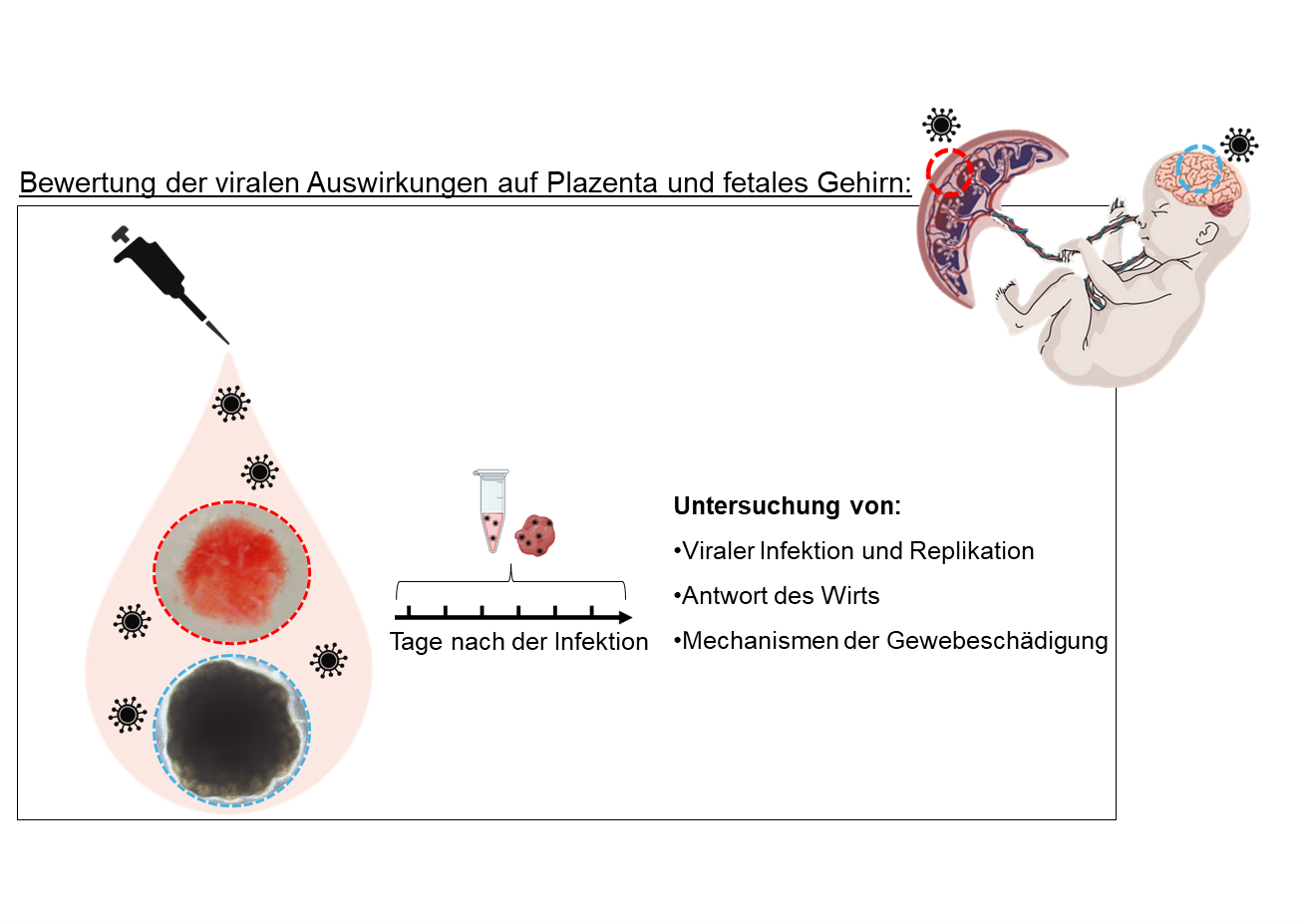
Virusinfektionen während der Schwangerschaft erhöhen das Risiko von Komplikationen sowohl für die Mutter als auch für den Fötus: Viren können Krankheitssymptome verschlimmern, zu mehr Fehlgeburten führen und die Entwicklung insbesondere des Gehirns beeinträchtigen. Wenn bei schwangeren Frauen eine Virämie auftritt und Viren ins Blut gelangen, kann dies die lebenswichtige Barrierefunktion der Plazenta beeinträchtigen. Das Virus kann dann die Plazentaschranke überwinden, die Gesundheit des Fötus gefährden und zu Entwicklungsstörungen führen, unter anderem im Gehirn. Zum besseren Verständnis der beteiligten Mechanismen braucht es humane In-vitro-Modelle, die Virusinfektionen im Plazenta- und Hirngewebe des Fötus möglichst genau nachbilden. Wir untersuchen mit komplexen Modellen, zum Beispiel mit Explantaten der menschlichen Plazenta und Organoiden des Gehirns, Infektionen mit SARS-CoV-2, dem West-Nil-Virus (WNV) und dem Zika-Virus (ZIKV). Mit diesen Modellen konnten wir den zellulären Tropismus, die Virusausbreitung sowie die Reaktionen des Wirts auf die Infektion untersuchen.